
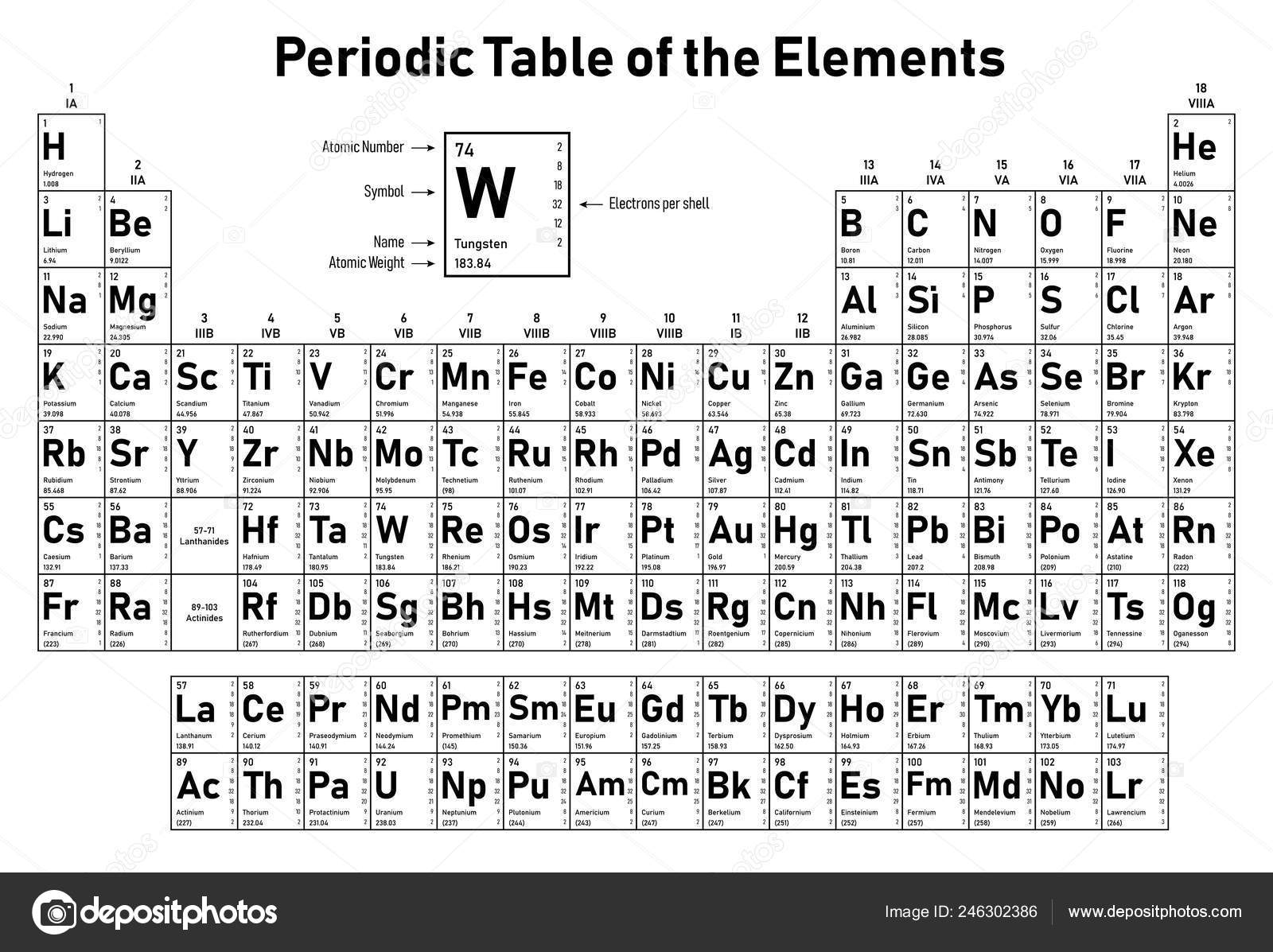
Elements that have atoms with similar outer shell electrical structures form groups. The horizontal rows are known as periods in this version of a periodic table, while the vertical columns are known as groups. The extended version of the periodic table is the current form that is widely used around the world. A new periodic table based on modern periodic law was constructed by upgrading Mendeleev’s periodic table. A number of artificial components were developed by scientists. The chemistry of these elements grabbed people’s interest as a result of this parallel. Chemists found a resemblance between the 94 naturally occurring chemical elements after learning about the periodic law. After learning about the fundamental unit of elements, scientists now had a thorough understanding of quantum numbers and the electronic configuration of elements in the periodic table. The number of electrons or protons in a neutral atom is equal to the atomic number. Physical and chemical properties of elements are periodic functions of atomic numbers. This resulted in the revision of Mendeleev’s periodic law, now known as contemporary periodic law. The advancement of quantum theory and the development of many atomic models revealed that the atomic number is the most fundamental attribute of a chemical element. Back then, scientists had no idea regarding the fundamental structure of the atom. In the late 18th century, Mendeleev devised his periodic table. The contemporary periodic table is based on Mendeleev’s periodic rule and the periodic table. Mendeleev organized the elements into a table, which became known as Mendeleev’s Periodic Table. A periodic function is one that repeats itself after a given time interval. Elements’ chemical and physical characteristics are a periodic function of their atomic masses. He made this finding in the form of the following statement in 1869, which is known as Mendeleev’s Periodic Law. When he arranged the elements in increasing order of atomic mass, he discovered that elements with comparable qualities appear on a regular basis. Mendeleev investigated the properties of all 63 elements known at the time, as well as their compounds. Mendeleev’s Periodic Law and Periodic Table It was because of its resemblance to musical notes, in which every eighth note is a repetition of the first note. It was dubbed the Law of Octaves by Newlands. He noticed that every eighth element shared attributes with the initial element. John Alexander Newlands, an English chemist, organized the elements in increasing order of their atomic weights in 1864. When atoms are grouped in increasing atomic mass order, the atomic mass of the middle element is almost equal to the arithmetic mean of the other two, and its properties are intermediate between those of the other two, according to this law.

Dobereiner’s law of triads was proposed by him. The physical and chemical properties of all three constituents of a triad were similar.
Dobereiner, a German chemist, named groups of three elements each triad in 1829. Arithmetic Progression - Common difference and Nth term | Class 10 Maths.Area of a Triangle - Coordinate Geometry | Class 10 Maths.Class 10 RD Sharma Solutions- Chapter 2 Polynomials - Exercise 2.1 | Set 2.Electric Potential and Potential Difference.Euclid's Division Algorithm - Real Numbers | Class 10 Maths.Step deviation Method for Finding the Mean with Examples.Theorem - The tangent at any point of a circle is perpendicular to the radius through the point of contact - Circles | Class 10 Maths.Concave and Convex Mirrors - Ray Diagrams, Image Formation, Applications.Section formula – Internal and External Division | Coordinate Geometry.ISRO CS Syllabus for Scientist/Engineer Exam.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.
